KatarzynaBialasiewicz/iStock via Getty Images
Investment Overview
Durham, N.C.,-based Humacyte (NASDAQ:HUMA) – a specialist in the field of regenerative medicine technology, announced its Q1 2024 earnings on May 10, triggering an impressive bull run on its stock that has seen the share price jump from $4.50 to $6.50 at the time of writing.
Prior to that, Humacyte stock had climbed from ~$3 per share to $4.50 per share between mid-April and its earnings announcement – in a little over one month, the stock price is up >110%. The company’s market cap valuation at the time of writing is ~$765m.
Humacyte achieved its Nasdaq listing in 2021 via a merger with the Special Purpose Acquisition Company (“SPAC”) Alpha Healthcare Acquisition Corp. As a reminder, according to Harvard Business Review:
A SPAC is a publicly traded corporation with a two-year life span formed with the sole purpose of effecting a merger, or “combination,” with a privately held business to enable it to go public.
SPACs raise money largely from public-equity investors and have the potential to derisk and shorten the IPO process for their target companies, often offering them better terms than a traditional IPO would.
Humacyte was able to raise ~$275m via its merger with Alpha Healthcare Acquisition Corp. The SPAC traded at $10 per share pre-merger, so even after recent gains we can see that Humacyte has yet to deliver for its initial investors, although, having fallen <$2 per share in November last year, recent progress has been highly encouraging.
In this post I’ll take a deeper dive look at the recent earnings and business updates, discuss the reasons for Humacyte’s sensational recent stock price gains, and speculate about whether the momentum is sustainable, likely to be checked, or perhaps faces a reversal in fortunes.
Humacyte – BLA Acceptance, Earnings and Business Updates
Humacyte’s focus is “off-the-shelf, universally implantable, bioengineered human tissues, advanced tissue constructs and organ systems”, according to its Q1 2024 quarterly report / 10Q submission, which also states:
We are initially using our proprietary, scientific technology platform to engineer and manufacture Human Acellular Vessels, or HAVs.
Our investigational HAVs are designed to be easily implanted into any patient without inducing a foreign body response or leading to immune rejection. We are developing a portfolio, or “cabinet,” of HAVs with varying diameters and lengths. The HAV cabinet would initially target the vascular repair, reconstruction and replacement market, including vascular trauma, arteriovenous (“AV”) access for hemodialysis, and peripheral artery disease (“PAD”).
We are also developing the HAV for coronary artery bypass grafting (“CABG”) and pediatric heart surgery. Over the longer term, we are developing our HAV for the delivery of cellular therapies, including pancreatic islet cell transplantation to treat Type 1 diabetes (our BioVascular PancreasTM or “BVP”).
Humacyte has had a Biologics License Application (“BLA”) accepted by the Food and Drug Agency (“FDA”) for its HAV targeting vascular trauma, and the FDA has agreed to an Aug. 10 Prescription Drug User Fee Act (“PDUFA”) date – the date by which it will announce whether the product will be approved for commercial sale and use, or whether it has been rejected, with the reasons for rejection outlined in a Complete Response Letter (“CRL”).
When the BLA was accepted in early February, it coincided with a spike in Humacyte’s share price from ~$3, to ~$5, although the gains were quickly eroded, possibly due to a $40m funding round completed at the end of the month. The FDA has agreed to a “priority review” of the application, shortening the review time from the usual ten months, to just six months.
The BLA – according to a Humacyte press release – is supported by:
Positive results from the V005 Phase 2/3 clinical trial, as well as real-world evidence from the treatment of wartime injuries in Ukraine under a Humanitarian Aid Program supported by the FDA. The HAV was observed to have higher rates of patency (blood flow), and lower rates of amputation and infection, as compared to historic synthetic graft benchmarks.
The HAV can be produced at commercial scale in Humacyte’s existing manufacturing facilities, which are expected to have the capacity to provide thousands of vessels for treating patients in need. The HAV has accumulated more than 1,200 patient-years of experience worldwide in a series of clinical trials in multiple indications, including vascular trauma repair, arteriovenous access for hemodialysis, and peripheral artery disease.
Announcing Q1 2024 earnings, Humacyte revealed a cash position of $115.5m, no revenues, R&D expenses of $21.3m, G&A expenses of $5.3m, and an overall net loss of $(31.9m), compared to $(37m) in the prior year period. In summary, although there are no immediate concerns, Humacyte’s current funding runway is likely to be exhausted by the middle of next year, hence the importance of the August PDUFA date.
Updating the market in last week’s earnings press release, Humacyte revealed the FDA had completed its pre-licensing inspection of its manufacturing facilities in Durham and that it had completed a Budget Impact Model “illustrating the potential economic value of the HAV compared to current standard of care in vascular trauma.”
Management expressed its confidence that the BLA will be approved, opening the door for a first commercial product launch – the first of many, management believes.
Mechanism Of Action, Competition
As shown below, Humacyte’s approach to regenerative medicine is innovative and somewhat unique – the company points out in its 2023 annual report / 10K submission that:
Despite the magnitude and critical nature of the diseases and conditions we are targeting, no significant advances in the open surgical market have been made in the last 35 years, and current treatment and products used in vascular repair, reconstruction and replacement suffer from various drawbacks
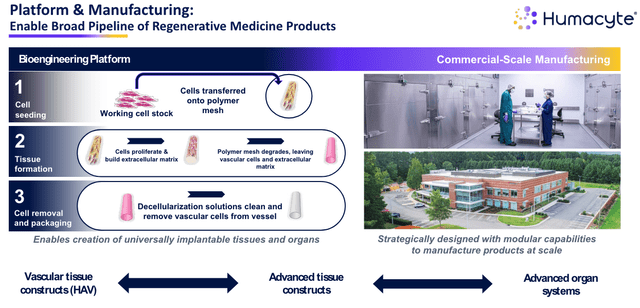
Humacyte approach to regenerative medicine (investor presentation)
A degradable polymer mesh allows for cell proliferation and the rebuilding of the extracellular matrix, while the product can be manufactured at scale at Humacyte’s plant – shelf life is ~18 months. The fact that it’s the patient’s own cells that are repopulating limits the risk of infection, and off-target damage to healthy cells.
Current standard of care treatments include harvesting of autologous veins, but this can be time consuming, expensive, requiring long stays in hospital, with mixed success rates, and not appropriate in all patient’s cases. Cryopreserved blood vessels and animal blood vessels are also used, albeit rarely, as the time taken to thaw restricts use.
Synthetic grafts are another type of treatment but come with a heightened risk of infection, Humacyte points out, particularly graft infections. In its pivotal, 69-patient V005 clinical trial, and in its humanitarian program conducted in Ukraine, HAV outperformed synthetic graft, as shown below:
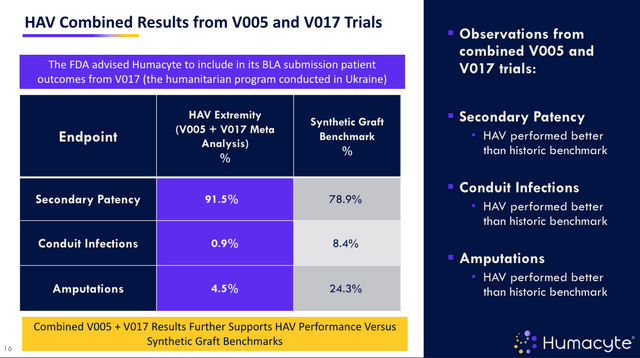
pivotal study data in vascular (presentation)
Although not by any means an expert in vascular wound treatment, the benefits of Humacyte’s approach strike me as relatively clear and obvious. However, that did lead me to question why more companies were not working on such an approach and whether Humacyte is able to patent protect its products?
The answers are that there are other companies developing HAVs – Humacyte mentions NovaHep AB, Xeltis AG, Hancock Jaffe, and Vascudyne Inc as direct competitors, and also mentions “bioabsorbable polymetric implants and electrospun or 3D printed tubular conduits” as potential rivals, noting that its commercial opportunity will be restricted by the success of any of these companies or approaches. As for patent protection, Humacyte states in its latest quarterly report:
As of December 31, 2023, our patent estate is comprised of 18 families of patents. Of these families, 14 are solely owned by Humacyte, one is jointly owned by Humacyte and Global Life Sciences Solutions USA LLC, one is jointly owned by Humacyte and Yale University, one is exclusively licensed to Humacyte from Duke University and one is exclusively licensed to Humacyte from Yale University.
Many of these patents and patent applications generally relate to the scaffolds used to make our vessels, the composition of our vessels, and systems and methods of manufacturing our vessels. Excluding any patent term adjustment or patent term extension, the U.S. patent relating to the scaffold used to make our vessels expires in 2032, the U.S. patents relating to the composition of our vessels expire in 2032 and the U.S. patents relating to the systems and methods of manufacturing our vessels expires in 2032.
To summarize, it seems that Humacyte enjoys a slight advantage in the race to a first approval for this type of technology – and patent protection is in place into the next decade, which is good news for the company, whose CEO, Laura Niklason, M.D., Ph.D., helped to develop the technology in the first place, at Duke University, and formerly, Massachusetts Institute of Technology.
Market Opportunity
Around the time of its IPO, Humacyte management speculated it could achieve peak annual sales in the double-digit billions, within a market worth ~$150bn. I doubt this is achievable, but the opportunities in play certainly warrant a rise in the share price if the FDA does approve Humacyte’s product in August.
Vascular trauma represents a small fraction of the overall opportunity, with only 200 level one trauma centers in the US at present. Management suggests that the Department of Defense would likely be a significant customer, stockpiling product for “deployment to sites of conflicts,” presumably needing to replenish supplies every 18 months, based on the shelf life.
To cover the domestic market, Humacyte estimates it needs only 20 sales representatives who will attempt to persuade surgeons that its product is a superior alternative to harvesting veins, a procedure most are unaccustomed to performing anyway. Nevertheless, a small sales team will take time to establish HAVs as a viable treatment option, and I’d speculate that the opportunity in trauma is in the low triple-digit millions, a figure that is apparently supported by analysts at HC Wainwright, who believe the product could achieve $205m revenues by 2031.
The vascular opportunity is – as mentioned previously – only the first of many potential follow-on opportunities, provided the product can show its mettle in a real-world setting. Arguably the most lucrative treatment settings, such as diabetes and cell therapy settings, are the furthest from being achieved, although given the recent rise of cell therapies, several of which are close to becoming “blockbuster” (>$1bn per annum) revenue products, the unmet need in cell therapy is clear, and the market opportunity substantial – likely even more so in diabetes.
Fresenius Agreement
Adding further value to Humacyte’s investment proposition is an agreement struck with Fresenius Medical Care, which operates kidney dialysis treatment centers. In 2018, Fresenius bought ~$150m of Humacyte convertible stock, and later invested a further $25m in the company.
In exchange, Humacyte:
Granted Fresenius Medical Care and its affiliates exclusive rights to develop outside the US and EU and commercialize outside of the US the Company’s 6 millimeter x 42 centimeter HAV and all improvements thereto, and modifications and derivatives thereof (including any changes to the length, diameter or configuration of the foregoing), for use in vascular creation, repair, replacement or construction, including renal replacement therapy for dialysis access, the treatment of peripheral artery disease, and the treatment of vascular trauma, but excluding coronary artery bypass graft, pediatric heart surgery, or adhering pancreatic islet cells onto the outer surface of the distribution product for use in diabetic patients.
Humacyte is obliged to pay Fresenius “a percentage of net sales in the low double digits” on sales made in the US, which in time falls to a single-digit percentage. In addition:
Fresenius Medical Care agreed to pay the Company initially, on a country-by-country basis for sales outside of the United States, the amount equal to the average cost of manufacturing the Company’s distribution product plus a fixed dollar amount per unit. Following a specified period, on a country-by-country basis outside of the United States, Fresenius Medical Care will pay the Company a fixed percentage of net sales for each unit sold in such country, such that the Company will receive more than half of such net sales.
The use of HAVs in dialysis can potentially help avoid infections by reducing use of catheters, hence Fresenius’ interest, and Humacyte is enrolling patients in a Phase 3 study designed to compare safety and efficacy with an autologous arteriovenous fistula in subjects with end stage renal disease. Phase 2 study results have hinted at HAV potentially being the superior option.
Concluding Thoughts: Recent Developments Support Share Price Gains – Near-Term Catalysts Support Further Gains
After a review of Humacyte’s technology, achievements to date, and opportunities – both short term and long – I come to the conclusion that the recent run-up in the company’s share price and valuation are well merited, that the vascular HAV appears to have a strong chance of being approved, and may compare favorably to current standards of care.
Innovative without being overly complex, readily available, and effective, HAVs appear to be a good solution to answering an unmet need, with plenty of opportunities in play for Humacyte to build upon a first approval – and a low triple-digit revenue opportunity – with several more approvals, including in collaboration with Fresenius, in end stage renal disease treatment, opening up a large and lucrative market, and bringing an experienced and global partner into play.
The level of competition Humacyte may face, and whether HAVs can ultimately supplant vein harvesting as standard of care, are the biggest challenges I would point to, alongside Humacyte’s limited funding and small sales force.
With an apparently significant first mover advantage, however, Humacyte strikes me as having a solid opportunity to establish a significant market share and adapt to the real world setting ahead of its rivals, and the support of the FDA, which have granted a priority review, is also reassuring.
As such, I make Humacyte stock a “buy” – even if it may take some time to establish commercial viability, and even if a $12bn revenue opportunity is wildly optimistic.
I could see Humacyte achieving a billion-dollar valuation within the next 12 months, however, and eventually, reporting a billion dollar annum revenue year, which ought to be substantially value enhancing.
My views by no means guarantee this will happen, and every investor should perform their own due diligence, but the billion-dollar market cap valuation, at least, strikes me as a likely, if not the likeliest, scenario, across the next 12 months.

